Do Genetics Affect the Way GLP1 Works for You?

If you’ve ever wondered why your friend lost 10 kilograms using Mounjaro, Zepbound, Foundayo, or Wegovy while you barely shifted the scale, science may finally have an answer, and it’s written in your DNA. Mounjaro and Wegovy are injectable medications containing specific active ingredients, such as semaglutide or tirzepatide, used for obesity and weight management. The active ingredient in these medications plays a key role in their effectiveness for weight loss and blood sugar control.
A major genome-wide association study published in Nature in April 2026 has uncovered compelling evidence that genetic variation plays a measurable role in how well GLP-1 receptor agonist medications work, and in how severe their side effects can be. The research, conducted by a team at the 23andMe Research Institute, analysed data from nearly 28,000 people and represents the largest study of its kind to date.
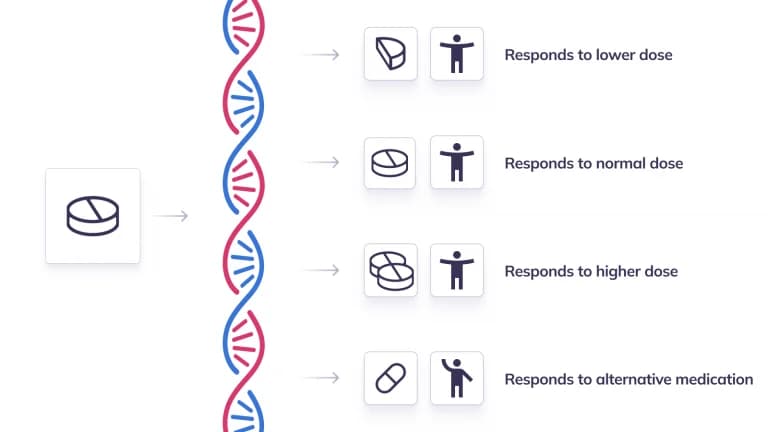
 Image of DNA strand known to affect GLP-1s
Image of DNA strand known to affect GLP-1s
The Weight-Loss Gap Is Real
GLP-1 receptor agonists, including blockbuster drugs like semaglutide (sold as Wegovy Injections or Wegovy Tablets) and tirzepatide (sold as Mounjaro or Zepbound, in the United States) have transformed obesity treatment over the past few years. But clinicians and patients alike have noticed a frustrating pattern: results vary enormously from person to person. Some patients achieve dramatic weight loss of 25 per cent or more of their body weight, while others lose very little or even gain weight on the same medication. Studies indicate average weight loss from GLP-1 receptor agonists can range from about 10% to nearly 20% of body weight, with some individuals achieving over 25% weight loss from baseline.
Until now, much of this variability has been attributed to factors like dosage, time on treatment, sex, age, and whether someone also has type 2 diabetes. These non-genetic factors are important and the study found they explain roughly 21 per cent of the variation in weight loss outcomes. However, some patients may even experience weight gain or less-than-expected weight loss, and higher doses or other factors such as genetics, lifestyle, and blood glucose control can influence these outcomes. But that still leaves the majority of the difference unexplained. The new research suggests genetics is part of the missing piece.
GLP-1 agonists are primarily used to manage blood sugar levels in people with Type 2 diabetes and can also aid in treating obesity.
How GLP-1 Agonists Work
GLP-1 agonists are a powerful class of medications that have changed the landscape of weight management and diabetes care. These drugs work by mimicking the action of glucagon-like peptide-1 (GLP-1), a natural hormone in the body that plays a key role in regulating blood sugar and appetite. By activating GLP-1 receptors, these medications help lower blood sugar, support weight loss, and improve overall health for people struggling with obesity or type 2 diabetes.
One of the main ways GLP-1 agonists help lower blood sugar is by encouraging the pancreas to release more insulin when it’s needed, especially after meals. This extra insulin helps move glucose out of the bloodstream, reducing blood sugar spikes. At the same time, GLP-1 agonists slow down gastric emptying which is the process by which food leaves the stomach so that glucose enters the bloodstream more gradually. This not only helps with blood sugar control but also increases feelings of fullness, making it easier to eat less and lose weight.
For many patients, GLP-1 agonists are an important part of a comprehensive treatment plan that includes dietary changes and other lifestyle adjustments. These medications are generally safe and well-tolerated, but like any drug, they can cause side effects. The most common issues are nausea, abdominal pain, and sometimes vomiting, especially when starting treatment or increasing the dose. It’s important to let your healthcare provider know about any adverse events or concerns, as they can help adjust your treatment to minimize discomfort.
The effectiveness and side effects of GLP-1 agonists can vary from person to person, depending on factors like age, body weight, personal and family history, and other health conditions. That’s why healthcare providers take a personalized approach, weighing the benefits and risks for each individual. While GLP-1 agonists can be highly effective for weight reduction and blood sugar control, they work best when combined with healthy eating and regular physical activity.
In summary, GLP-1 agonists offer a valuable option for people looking to manage their weight and lower blood sugar, especially those with obesity or type 2 diabetes. They are not miracle drugs, but when used as part of a thoughtful treatment plan, they can make a real difference in long-term health and weight management.
Two Key Genes in the Spotlight
The study zeroed in on two genes: GLP1R and GIPR. Both encode the receptors that these medications target, making them biologically intuitive candidates for influencing drug response.
The most significant finding involved a missense variant in the GLP1R gene (known as rs10305420). Individuals carrying the effect allele of this variant experienced an additional 0.76 kilograms of weight loss per copy of the allele compared to non-carriers. While that might sound modest on its own, genetic effects are additive and cumulative, and this is just one variant among what are likely many yet to be discovered.
The researchers believe this variant may work by altering the signal peptide of the GLP-1 receptor, potentially improving how the receptor is transported to the cell surface rather than changing how the drug binds to it. In other words, some people may simply have more of the receptor available for the drug to act on. These genetic differences can also affect insulin secretion and blood glucose control, which are central to the mechanism of GLP1-based therapies. Certain genetic profiles may be associated with a higher risk of adverse effects, such as nausea and vomiting, and may also influence blood glucose regulation.
A second finding concerned the GIPR gene. A well-characterised variant (rs1800437) was linked to an increased risk of nausea and vomiting, but only in people taking tirzepatide, which is a dual GLP-1 and GIP receptor agonist. This makes biological sense: the GIP component of tirzepatide is thought to help buffer the nausea caused by GLP-1 activity, and a partial loss-of-function variant in the GIP receptor could undermine that buffering effect. Strikingly, the study estimated that individuals carrying risk alleles at both loci had dramatically elevated odds of experiencing tirzepatide-related vomiting.
Efficacy and Side Effects Are Connected
One particularly interesting aspect of the research is the finding that the same genetic region that predicts better weight loss also predicts worse nausea and vomiting. Co-localisation analysis showed that the efficacy and side-effect signals in the GLP1R gene likely share the same underlying causal variant. This aligns with what many patients and clinicians have observed anecdotally: people who feel more nauseous on these drugs often tend to lose more weight.
Who Responds Best?
The study also confirmed several non-genetic patterns. Women tended to lose more weight than men. Tirzepatide was associated with greater weight loss than semaglutide over similar treatment periods. Efficacy decreased modestly with age and was lower in people who also had type 2 diabetes, hypertension, or fatty liver disease. High cholesterol and other weight-related health conditions, such as sleep apnea, can also influence eligibility and response to GLP-1 agonist treatment. According to the American Diabetes Association’s guidelines for obesity and diabetes management, GLP-1 agonists are recommended for patients with weight-related health conditions. GLP-1 receptor agonists may improve nonalcoholic fatty liver disease (NAFLD) and decrease joint pain by alleviating excess weight burden.
Ancestry also played a role, with participants of European descent showing the greatest average weight loss, followed by those of Latino heritage, and then African American participants. Notably, the key GLP1R variant is most common in people of European and Middle Eastern descent and least common in those of African descent, a pattern that may partially explain observed differences in drug response across populations.
What This Means for the Future of Treatment
The study’s authors argue that these findings lay the groundwork for a precision medicine approach to obesity treatment. If doctors could test for relevant genetic variants before prescribing a GLP-1 medication, they might be able to predict which drug is likely to work best for a given patient, how aggressively to escalate the dose, and how to manage anticipated side effects.
The researchers built combined models incorporating genetic and non-genetic factors, and showed that these models could stratify patients by likely weight loss outcomes, a proof of concept that could eventually translate into clinical tools.
The study highlighted that identifying predictive factors could help guide treatment strategy, including the choice of drug, dose, and speed of dose escalation. This is especially relevant given that new options are entering the market like Eli Lilly’s oral GLP-1 orforglipron (brand name Foundayo) which received FDA approval in the US in early April 2026; and the treatment landscape is only growing more complex.
When considering weight loss medication, it is important to use FDA or MHRA approved GLP-1 drugs, as these have been reviewed for safety, effectiveness, and quality. Some health care providers may prescribe GLP-1 medications off label for conditions other than those approved by the FDA or MHRA, but this should be done with careful consideration and patient guidance.
In the United States, there are significant risks associated with compounded semaglutide, compounded tirzepatide, and other compounded versions of GLP-1 drugs. Compounded GLP-1 drugs are not FDA approved, meaning the agency does not review them for safety, effectiveness, or quality before they are marketed. Compounding pharmacies producing these medications may lack proper regulatory oversight, increasing the risk of incorrect dosing, adverse events, and inconsistent efficacy.
The FDA has expressed concerns regarding unapproved versions of GLP-1 drugs, including semaglutide and tirzepatide, and has established an import alert to prevent GLP-1 active pharmaceutical ingredients with potential quality concerns from entering the U.S. supply chain. The FDA has also received reports of adverse events related to compounded versions of semaglutide and tirzepatide, including symptoms such as nausea, vomiting, diarrhea, abdominal pain, and constipation.
Health care professionals play a critical role in reporting adverse events, ensuring patient safety, and guiding appropriate prescribing practices for GLP-1 medications, especially when considering compounded or off-label use.
Summary
The science is still early and the genetic effect identified so far are modest, and the researchers themselves note that larger datasets will likely uncover additional variants and improve the predictive power of genetic models. But the direction is clear: your genes do influence how GLP-1 medications work for you, and the era of one-size-fits-all prescribing for these drugs may eventually give way to something far more personalised.
For now, if you’re taking a GLP-1 medication and not seeing the results you expected, or struggling with side effects, it’s worth knowing that biology may be playing a bigger role.
NowPatient has taken all reasonable steps to ensure that all material is factually accurate, complete, and current. However, the knowledge and experience of a qualified healthcare professional should always be sought after instead of using the information on this page. Before taking any drug, you should always speak to your doctor or another qualified healthcare provider.
The information provided here about medications is subject to change and is not meant to include all uses, precautions, warnings, directions, drug interactions, allergic reactions, or negative effects. The absence of warnings or other information for a particular medication does not imply that the medication or medication combination is appropriate for all patients or for all possible purposes.